Module 7: Quality Assurance
Materials in this module will help you:
- Understand the purpose of Quality Assurance, the role of the Quality Assurance Lead at your site and how you will support these activities
- Understand how test kits are ordered, stored and evaluated
- Support ordering and inventory management
- Perform positive/negative quality control testing of new kits
- Understand the ongoing quality assurance processes done at each site and the documentation required

Quality Assurance Involves:
- Training and updating of staff skills
- Monitoring of how test kits are ordered, received, stored and used
- Regular evaluation of test kits to make sure they are working properly
- Regular evaluation of the proficiency of staff and procedures at your site to ensure correct results
Your Responsibilities

Training
- Participate in new staff training and mentorship at your site.
- Complete a staff certification testing panel after your training is complete to fully qualify as a tester.
- Take part in refresher courses annually or after any prolonged absence.
- Keep track of the records of your training, and submit them to be stored in your HR file.
Test Kits and Processes
- Make sure that new orders of test kits are stored in the storage area away from existing kits; check to make sure they haven’t been frozen or damaged. If it is the practice at your site, tell the QA lead they have arrived.
- Record the temperature in the kit storage area daily; alert the QA lead if the temperature goes out of range or if the supply of tests seems low.
- Record the lot number and expiry date of every test you do in the daily log.
- Participate in positive/negative quality control testing of the kits as requested.
- Participate in the Proficiency Testing program operated by Institute for Quality Management in Healthcare (IQMH) to assess testing processes at your site. All counsellors participate.
- Report invalid/unexpected results to the QA Lead; record on the incident log as well as the daily log.
Invalid Test Results
When invalid tests happen, don’t discard the testing material (membrane, etc.). Take a picture and send it to the Ministry of Health (HIV and Hepatitis C Programs) for review.

Evaluating Kits with Positive/Negative Quality Controls
Your site has vials of positive and negative control material to regularly evaluate the kits you are using. Control testing should be done: when a new lot of kits first arrives at your site; if the temperature in your storage area falls out of range; AND monthly.
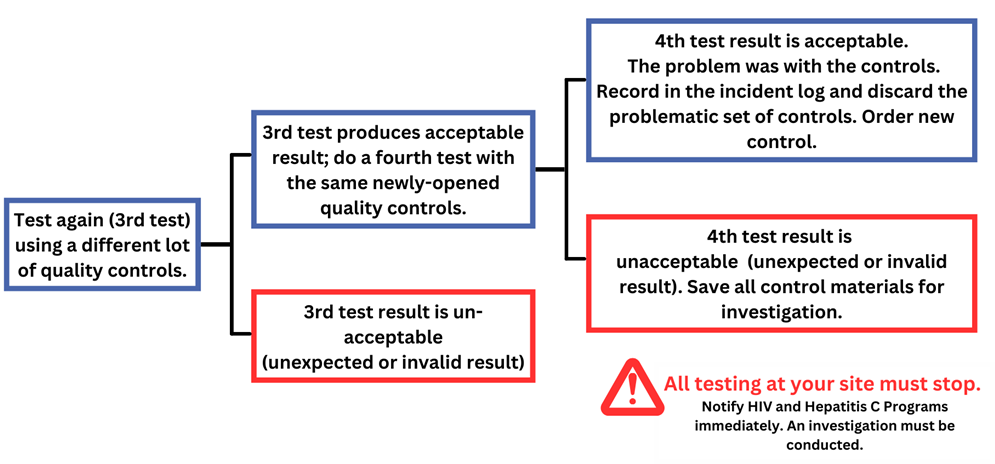
What if Quality Control Testing Fails?
Immediately:
- If the positive is not positive, or the negative is not negative, don’t discard the test material (membrane, etc.) Take a picture. Both may be used to investigate.
- Alert the Quality Assurance Lead and/or Supervisor.
- Record what happened in the incident log including the expected result, the result obtained and subsequent efforts to resolve the cause of this incident.
Second Test:
- Repeat the testing a second time using the same lot of test kits and the same positive/negative quality controls. Tests of each control should be done one at a time, not simultaneously.
- If the results are acceptable (as expected), record them in the quality control log and incident log. Testing at your site can continue as usual.
- Try to identify why the first test came out wrong (i.e. mix up of materials, missed a step) Record in the incident log.
How Testing Processes are Assessed at Your Site
Rapid testing sites are also expected to evaluate their staff and procedures regularly as follows:
| Parallel Testing | Every time a reactive test occurs, clients are advised to confirm the result with standard laboratory testing. This also helps ensure that testing is working well at your site. Report to your Quality Assurance Lead whenever follow-up testing on a reactive rapid test is non-reactive or when a negative sample submitted for standard testing in the window period has a reactive result. |
| Monthly Assessment Of Testing Results | Each month, your Quality Assurance Lead completes a monthly summary of testing at your site including the number of positive, negative and invalid tests done, the number referred to PHOL for further testing, any unanticipated results and kit usage. This information is gathered from the daily log. |
| Notify HIV and Hepatitis C Programs if: If greater than 1% of POC tests were falsely reactive in one month of testingIf greater than 1% of POC tests were invalid in one month of testing | |
| Proficiency Testing | The Ministry has established a regular Proficiency Testing Program for rapidtesting sites, administered by the Institute for Quality Management in Healthcare (IQMH). Your site will regularly receive unknown samples from IQMH to test and return for assessment. Each staff person who delivers client testing at your site must participate. Rotate staff as required.When the results have been analyzed they will be available through the secure Qview portal managed by IQMH. Your Quality Assurance Lead will have access. The Ministry monitors the outcomes of this testing, to ensure that errors are investigated and corrective action taken, as needed. |

Summary of Required Logs and Documentation
Daily Tools
- Rapid HIV Testing Record Daily Log – a daily log completed by all testing staff, showing the date, kit lots in use and all the rapid testing being done (reactive and non-reactive), as well as QC tests, and requests for HIV testing sent to PHOL.
- Environmental Monitoring Log – daily record of the temperature that kits are stored.
Monthly Assessment usually done by the Quality Assurance Lead at the Site
- Monthly Data Report – A spreadsheet updated on a monthly basis that summarizes kit usage at your site (including kits used for proficiency and certification testing, as well as damaged and invalid tests). The monthly log also records the number of tests sent to the Public Health Ontario laboratory and notes any discrepancies between rapid POC test findings and those produced by the Public Health lab. This summary helps assess quality assurance practices at your site, and generates the information that must be entered into the www.hivpoct.ca portal when ordering new kits.
Other Records kept at the Site and Updated as Activities Occur
- Quality Control Log – record of the QC tests done for regular supervisor review
- Incident Log – record of any discrepancies (including invalid tests, positive/negative control test errors, and proficiency test errors), the investigations done, and solutions identified
- Proficiency Testing Records – The outcomes of individual proficiency testing
- Records of Counsellor Training and Validation – Maintained and updated in HR files
- Material Safety Data Sheets (MSDS)
- MSDS are available for the HIV POC tests https://hivtestingontario.ca/poc/kit-storage/
Having MSDS available at your site is a Workplace Hazardous Materials Information System requirement.
Resources for More Information
- Materials on the Ontario HIV Testing Program web site www.ohtn.on.ca/hivtesting, which include quality assurance templates for download https://hivtestingontario.ca/poc/quality-assurance-templates/
- Quick Reference Guide to Ordering Supplies https://hivtestingontario.ca/wp-content/uploads/2020/06/HIV_POC_Testing_Supplies_QuickReferenceChart.pdf
- The package inserts for the HIV-1 Antibody Test Kit Controls and the INSTI HIV-1/HIV-2Antibody Test Kit.